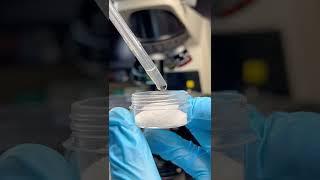
Salt-water Versus bacteria under the Microscope!
Комментарии:

try salt and demon.
Ответить
And this is why people used to put lots of salt on the meet.
Ответить
Add urine in virus sample
Ответить
Then how is the ocean full of bacteria. Or is it because the salt is way less concentrated? 😊
Ответить
Celtic salt please
Ответить
a genocide every video
Ответить
Quick, everyone snort salt. It's flu season
Ответить
Try copper under the microscope
Ответить
Harpic
Ответить
Bacteria:5 minutes ago,i lost 50k bacteria in the blink of an eye.
Ответить
I love putting salt in bacteria
Ответить
Sanitizer vs dirty bacteria hand please 😊😊😊
Ответить
SALT BAE IS A LEGEND
Ответить
Saline solution is way better than commercial mouthwash
Ответить
But if it cleans the water we drink is allowed.
Ответить
I would like to see if this kind of bacteria from video would survive or even thrive in hypotonic solution by completely deionized water (100% water, devoid of electrolites).
Or if you could do similar video about extremophilic microorganisms with similar comparison to "normal/ordinary" microorganisms.

What have you just seen is hypertonic osmotic solution that has way higher Sodium and Chlorine ions in solution than in the medium that bacteria is in.
In cells, every single molecule or atoms (like simple electrolites like Na, K, Mg, Ca, Cl, HCO3, NO2 and etc) concentration must be in delicate balance betweeen extracellular(outside of cell) and intracellular(inside of cell, or simply plasma) concentrations, which some molecules are more abundant outside of cell, and some more abundant inside.
Given this sudden gradient change in concentration caused by hypertonic solution by salt(NaCl), this acts as double whammy.
First, cells gets dehydrated due the fact cells have natural mechanism that exchanges the ions of Ca and K, in exchange for Na, at the cost of energy in form of ATP, which gets hydrolyzed at the cost of water indirectly (this is necessary because the cells needs to maintain the cell integrity, if it gets too large or small, cell risks spontaneous necrosis), but that's only 1% of total loss of water, its more in K ions loss due to higher Na levels.
Second is more severe and is more of aspects of simple physics of osmotic pressure, or in another words simple osmosis. Everything in solution is aspiring(or tends) to get to a perfect balance or equilibrium, which means every part of solution must gets even or equal in concentration. And since the concentration of salts inside and outside of cells are different, the extreme rise in salt concentration results in cells to be "crushed"(actually shrivel due to water exiting from cell), and in change of ionic strength, all enzyme gets denatured and losing the essential functions that keep cells in life.
That effect can be compared to what would happen to us when we would be in an environment where air pressure is at least 100 times higher than that of our planet.
The opposite effect happens with hypotonic osmotic solution, where completely pure water(without any electrolites or other particles present in it). In that case, water would just charge in the cells, blow them up like ballons until they pop.
That's what would happen to us if we would be in space where near vacuum environment is without astronaut suit, water from our cells would just boil out from us due to low pressure.

Apple cider vinegar test please and bayleaf oil or water
Ответить
What happens iff you put alcohol at the bactira
Ответить
That's why we pickle food.
Ответить
Do human saliva to see how many germs are in the mouth. 1st: When you get up and before you brush, then after. OIL PULLING with coconut VS TOOTHPASTE.
Ответить
Try bleach under the microscope vs bacteria.
Ответить
That's why Soils are saline bcz of salty water applications
And baldness also reason

Lemon juice on bacteria please
Ответить
Try gasoline
Ответить
Honey
Ответить
This is what happens every time you take a bath use sanitizer or clean any thing!!!
Ответить
This is genocide.... MURDERER!!!😂
Ответить
I don't know if I quit eating salt or start eating it
Ответить
But we all know that there is bacteria that prefer extreme saline conditions and those that can tolerant and those that can't leave in such conditions...
Ответить
So, is there no bacteria in the Ocean?
Ответить
Can you try with onion or garlic Thank you
Ответить
Fire on microscopic
Ответить
Germnicide.
Ответить
Bacteria are so cool and fascinating
Ответить
So salt = safe 🤔
Ответить
Salt enemy of bacteria 😂😊
Ответить
Use turmeric next time
Ответить
So Cool! Makes me wonder if this is relevant for viruses...or just bacteria?
Ответить
Cool, you find it cool huh, stop this hate against the bacteria they are alive!! SAVE THE BACTERIA , BACTERIA MATTERS YAY!!!
Ответить
Ah yes, classic mass genocide
Ответить
I feel like we're just bacteria on this planet, just waiting for a higher being to massacre us for content in part of some greater world that we can't see because we're so tiny.
Ответить
hello can you try Rosemary leaves please
Ответить
Kiling for content😂
Ответить
Can you try blood vs bacteria
Ответить
Please
Ответить
Can you try sewerage water
Ответить
murder
Ответить
Sperm and ovum plz ❤
Ответить
I wanna see clorox
Ответить