Making Potassium with Homemade Metal Convertor
Комментарии:

You got argon going in but nowhere for it to come out, so it can't possibly fill 100%
Ответить
Very nice.
Though does argon purging really work when the reaction vessel only has one opening?
Perhaps a tire-pump vacuum pump could evacuate the system enough to effectively remove the oxygen before adding argon?

This was a fun video. I can't wait to see the rest of the metals you make with your pipes.
Ответить
Love this retort!
Was there a reason you didn't put mineral oil in the collection flask?

At that size I wonder if one of those little electric induction heaters would do the job more easily and controllable?
Ответить
Great video, just came across your channel. Do a very great job explaining everyrhing your doing.
Ответить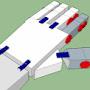
I wonder, if you get this up to ~1100'c could you use iron or copper to extract magnesium metal from it's oxide/chlorides?
Ответить
what is the furnace you are using at the end?
Ответить
Nice design! Pros for flange setup. As for flanges, you need some kind of seal between them, otherwise it will always leak. Use foil made from not reactive soft metal, like copper, tightening of screws will squeeze it and should seal flange. Such copper seal rings are used in high vacuum setups.
Also, like other people noted, reactor was definitely not flushed with argon. Without second port on flange, vacuum is one choice, and other would be small diameter tube that fits in "air condenser" so you put argon directly into reactor, and then it is pushed out through space between both tubes. Of course such gassing tube would be removed before distillation.

My uncle in alabama is making stuff like this also. He's got a set like this in the woods cause he says some experiments can create toxic gas. He must like doing these experiments cause he's up all night long doing them.
Ответить
Love how nice and shiny the potassium was when made, nice job there! 👍👍 Potassium is by far one of my favorite elements due to it being one of the most common natural sources of radioactivity and antimatter! Yes, you read that right antimatter as potassium-40 sometimes decays to argon-40 via positron emission! ☢☢
Ответить
Could you provide links to the parts or list the part sizes?
Ответить
CaCl ???
Ответить
Your video is awesome 👍
Ответить
CaCl₂
Ответить
Have you tried this with magnesium? Aluminum ? Easier to get than calcium metal.
Ответить
Bro you sound like your 14
Ответить
Great channel I love your enthusiasm and passion.
Ответить
...excellent video, ty
Ответить
Can you please run a test tobsee if you can make potassium or sodium from aluminium powder? Magnesium powder is really hard to get in Europe
Ответить
Advanced Thinkering made Potassium und Caesium with his vacuum system.
Ответить