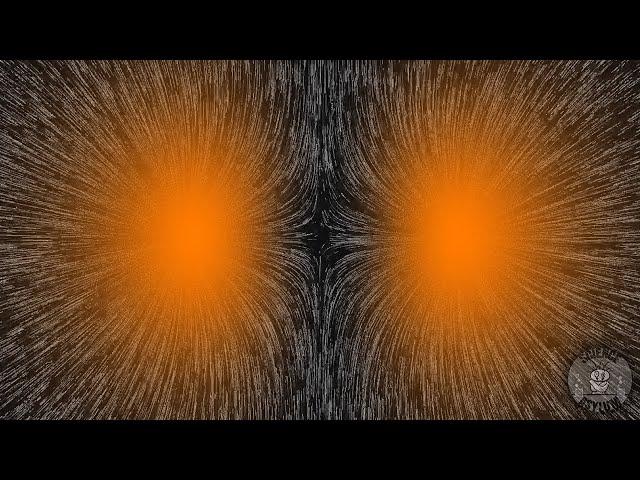
Atomic Orbitals, Visualized Dynamically
Комментарии:

Is an electron the only negative magnetic monopole ?
I would presume the proton is not a positive magnetic monopole because it is comprise of a particle that has a negative 1/3 particle so it, itself is a bipolar magnet, right? Only the positron would be a positive mono pole magnet, wouldn't it?

Wpuld string theory replace the electron cloud model
Ответить
I wish you would have applied this to a few different elements.
Ответить
i' am hurgy
Ответить
Grate explanation
Ответить
It's cats, all the way down.
Ответить
This is by far my favorite video about atomic orbitals! Thank you so much for the explanation! I wish I’d found this in the beginning of my studies
Ответить
When I was learning this in college, I got really hung up on the idea of the wavefunction telling us where the electron was likely to be. Where we would most likely find it. Apart from hydrogen atoms, there are multiple electrons, and certainly there are many electrons in the universe; what a detector can tell you is that it found an electron. They are all identical. How can we possibly know that this cloud of electron orbital (or any other electron wavefunction, for that matter) is describing the behavior of a single particle if there are trillions of identical particles all over the place?
Note that this is entirely a different matter from the QM treatment of multiple particle wavefunctions. I'm aware of how that all works mathematically. My confusion was perhaps best illustrated by the interpretation of quantum leaps and tunneling. When we fire an electron at a barrier and then detect an electron on the other side, what makes us think that it's the same one? It would be simpler if the barrier absorbed the original and then emitted a different one on the other end. The barrier has tons of electrons in it.
Anyway, I've long since moved on, but I do wish someone had taken the time to explain that to me.

Hmm. And the atoms in my cake?
Ответить
حدانا بيقولو توماتيكى توماتيكىالرياضة بقى يعنى جووول هههه بس
Ответить
Intetesting! What do the orbitals look like for atoms that undergo Cooper pairing?
Ответить
I wonder if he talks that way really, and if so, why.
Ответить
The cake is real!
Ответить
"it's okay to be a lil crazy"✨
Ответить
I always dreamed that physics and chemistry would later unify everything measure everything on a table or chart and just get rid of probability. The problem is the people that go into these fields love math. They turn everything into an equation. Thus, we are still dealing with probability.
Ответить
It’s Brownie, not Cake
Ответить
Wow. I have to buy your book despite that I am only software engineer
Ответить
i think the cake is not a lie
Ответить
Vhat
Ответить
How fast are electrons moving around atoms?
Ответить
One thing about orbitals that strike me as strange is the fact that they aren't completely spherical. Why is it that they align with some axis? Doesn't the electron stay attached to the nucleus because of the electric field around the nucleus? And isn't this field the same at any point around the nucleus? Why would the electron "prefer" one place over another?
Ответить
Is the 'cake is a lie' reference from portal the video game? Or is it just an established science joke that existed even before portal? In that case, what does it even mean?
Ответить
Thanks for reminding me to introduce Portal to my kid.
Ответить
What would the ith period look like, do we add aa double-Lanthanide expansion for the g shell?
Ответить
The cake is made of atoms,so one could say that everything made of atoms is made of cake,if cake was your only example,lol,love your video,where can I get your book?
Ответить
I'm doing last minute studying for chemistry right now and this was a huge help. Thank you!
Ответить
Cake is NEVER a lie.....unless it's vanilla.
Ответить
Dumbbells and bagels!!!😂😂😂
Ответить
influence of potato
Ответить
I love your channel, it makes things easy to understand for people without a background, tysm
Ответить
I always thought electron orbitals were orbits, today I learned they are not orbits.
Ответить
hi how does phase play into this? like we learn in school that, p orbital for example has opposite phase lobes on either side of its axis. from what ive found the phases represent the curve of the wave? and play a part in lcao, but where does that come in this video's explanation of orbitals?
Ответить
As USA wants more young people get into STEM, why still keep the same style of the 70s. A scientist is not equivalent to nerd. Why do you always make the look of a nervous nerd. It is absolutely no need for that, just act naturally.
Ответить
superb content
Ответить
Someone aligns with the Railroad rather than the BOS.
Ответить
How can electrons absorb photons? I thought the wavelength of light was bigger than an atom? So when light reflects off of an object, what is it reflecting from? I think I may be more than a little crazy ...
Ответить
"Visualize" is the word since orbitals have no appearance. They lack the basic requirements for being seeable and, in general, ubiety.
Ответить
So where were the moving dynamic pictures? the electron at least the free electron is a infinitely small point object.
Ответить
What causes electrons (particles with mass) to follow the crazy path that they must, within an atom?
Ответить
this is another helpful visual to imagine what atoms really look like. Thanks! For those who may hate dot structures just want to say the classic diagrams are DIAGRAMS.they are a tool , no one is claiming that a diagram is what something really looks like. Think of a blueprint, no one looks at it and rages at an architect that THAT'S NOT WHAT A HOUSE REALLY LOOKS LIKE!
Ответить
I feel it's easier if you say that an orbital is a region of space where you may find an electron
Ответить
I prefer Pi to cake.
Ответить
Electrons be like velcro
Ответить
The cake is a lie, NIIIIIICE!!! •Shout out!!!
Ответить
"The cake is a lie" takes me back to my fun teenage years when gun went pew instead of math math chem chem
Ответить
Fascinating
Ответить
THE CAKE IS A LIE!!
Ответить
the relationship between the wavelength of light and the minimum object size resolvable would be a nice topic for a video IMO
Ответить
stop talking like socially awkward high school student. cringe.
Ответить