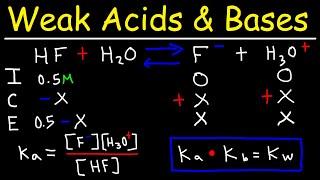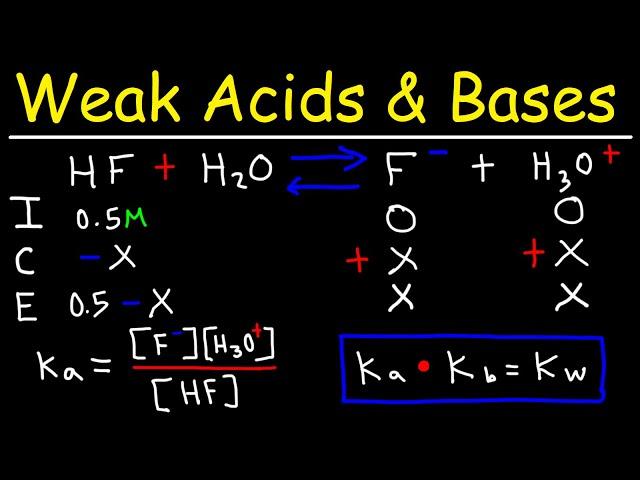
pH of Weak Acids and Bases - Percent Ionization - Ka & Kb
Комментарии:

I want to ask something because i don't understand. What if the Ka is not small enough to ignore the x? how to change it into ax2 + bx + c = 0?
Ответить
sure as hell, you were called for this.
Ответить
How can you say that ph and poh is difference even though putting same concentration in same -ve logarithms??
Ответить
Awsome
Ответить
I love you bro
Ответить
One love no homo
Ответить
I don’t even study I just come watch your videos 10 minutes before my quizzes and get 100% 😂
Ответить
Awesome video with so many examples with different situations ❤❤❤
Ответить
Very interesting ❤
Ответить
Please how do you know it's a strong or a weak acid and weak or strong base.
Ответить
Thank you
Ответить
Thank you ❣️ so much 🥰 it has been long time i studied this topic acid and bases and when I reach at weak acid I'm always confused 🤔 but today i found this video 📸 and i understand easier, may the Lord 🙏 bless you 💯💯💯😊
Ответить
I'm so glad to have you
The way you explained the ka,kb and percent ionization is explicit

I didn't study anything I just went through your videos in a short period I acquired more knowledge ad passed my test👋👋u are the best ever
Ответить
i was a fan until you said cross multiply. such a weak term.
Ответить
I still don’t get why “0.75-X” was changed to “0.75”. I don’t understand what is negligible that you said
Ответить
I need to calculate how much quantity of Conc H2SO4 Will be required for a 100 mL solution to reduce the pH from 6.5 to 4.5. How do I calculate it ??
Ответить
Sir how do you got 1.310^_11
Ответить
Woah.. Question 3 was very difficult..
Ответить
Could you please make a video for solving the Systematic Treatment of Equilibrium?
Ответить
when will the Equilibrium shift to the left instead of the right in the ice table
Ответить
How do you find Ka if given hydronium equilibrium concentration and told acid intial concentration
Ответить
Me: Learning this after the Exam 🤦♂️
Ответить
እናመሰግናለን
Ответить
Thanks very much for these videos😍, but I didn’t understand why sometimes we use Ka and other times we use Kb ?
Ответить
I will never understand, as going for a BA for mechanical engineering, computer science, and even physical therapy, we are required to learn all these concepts of chemistry! Do they teach chemistry majors engineering math? Or computer coding? Or other non related classes to those wanting to be chemists? Why is chemistry so important EVERY other STEM major must learn it? I will NEVER use these concepts!
But thank you for these videos, only way I'm getting through chemistry 2 just so I can forget it forever.

Your videos are not playing off late. Kindly check on them
Ответить
bro is responsible for my grade
Ответить
Hey, I’d like to ask about Cl. Why didn’t we include it in the reaction ?
Ответить
can someone help me?
A saturated aqueous solution of salicylic acid has a concentration of 0.018 m at 25 °C.
Calculate the pH of the saturated salicylic acid solution.
how do i calculate this and how do i find the ka if its not included.

Once upon a time JG
Ответить
Your absolutely a genius SIR. Salute from another continent.
Ответить
Mr man thanks 👍 alot always being blessed by God
Ответить
at 16.38 what will happen to Cl atoms ? didn't the problem states NH4Cl, so why only use NH4?
Ответить
L for this one. You did not explain correctly
Ответить
Sir, for the 2nd exercise when we were fitting the value for [NH3] why didn`t we put (0.25-x)
Ответить
I thought I was doing chemistry not mathematics
Ответить
thank you
Ответить
thx alot 🙏
Ответить
You are legit a legend. You’ve helped me get through Chem 2 when the teacher didn’t even give us any lectures. Triple thumbs up Brodie👍🏼👍🏼👍🏼
Ответить
Thanks a million for this video. But you didn't explain why Na in NaF is a spectator ion
Ответить
THANK YOU SO MUCH
Ответить
fkchemistry..:)
Ответить
what happened to the chloride in question 3?
Ответить
For the 3rd question, why can’t we use NH3 + HCl —> NH4+ + Cl-
Ответить
I was struggling with this concept in my chemistry class but this video cleared all of it up in under 30min! I really appreciate it!
Ответить